Chemicals
Ammonium Buffer PH10
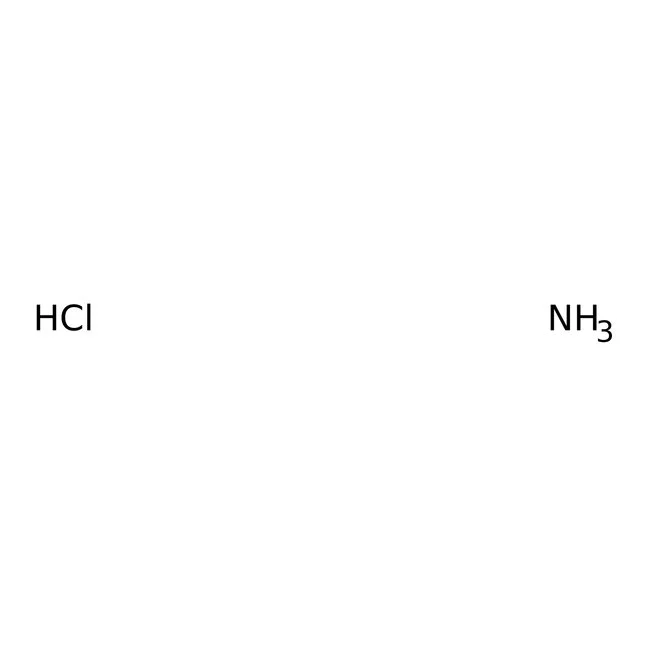
The buffer is a mixture of ammonia (NH3) and ammonium (NH4+). … Since ammonia (NH3) is a weak base, it will have a pH above 7 and since ammonium(NH4+) is a weak acid, it will have a pH below 7. Ammonia (NH3) is a weak base and ammonium (NH4+) is a weak acid. Ammonia (NH3) is the conjugate base of ammonium (NH4+).
KSh5,670.00
The buffer is a mixture of ammonia (NH3) and ammonium (NH4+). … Since ammonia (NH3) is a weak base, it will have a pH above 7 and since ammonium(NH4+) is a weak acid, it will have a pH below 7. Ammonia (NH3) is a weak base and ammonium (NH4+) is a weak acid. Ammonia (NH3) is the conjugate base of ammonium (NH4+).




There are no reviews yet.